Latest Blogs
Spinocerebellar Ataxia: Types, Symptoms, Causes, Diagnosis and Treatment
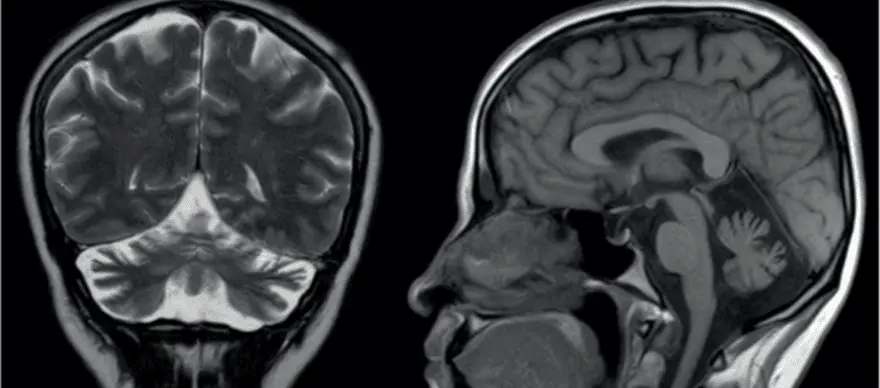
What is Spinocerebellar Ataxia Spinocerebellar Ataxia (SCA) or Spinocerebellar Degeneration is a neurodegenerative disorder that affects the proper functioning of brain-body coordination and may cause difficulties with walking and performing fine motor movements, swallowing, and weakness in muscles controlling the eye movement. Numerous conditions can cause Cerebellar and Spinocerebellar Degeneration. However, acute injuries or infections and damages affecting the parts of the brain controlling the motor movements (cerebellum) are the significant cause of Spinocerebellar Ataxia. Currently, there is no effective cure or treatment for Spinocerebellar Ataxia. However, not all ataxia types cause equally severe abnormalities. In general, treatments for most Spinocerebellar Ataxia cases involve focusing on relieving the specific symptoms and lowering the progression of ataxia. Types Of Ataxia There are several types of ataxia. Based on their symptoms and their causing factor for developing neurodegenerative conditions, researchers and doctors have classified ataxia into numerous types, some of which are given below. Knowing the types and their unique symptoms can help you and your doctors evaluate and rule out similar disease conditions and determine a proper treatment plan. Ataxia-telangiectasia: Also known as Louis-Bar Syndrome, it is a rare type of early onset inherit ataxia (passed from parents to children) that affects the immune and nervous systems of the body. The symptoms of ataxia-telangiectasia can develop at an early age, usually at 1-5 years of childhood, and its symptoms tend to get worse with time. Friedreich’s ataxia: Friedreich’s ataxia is the most common type of ataxia affecting people worldwide. Although both Friedreich’s ataxia and telangiectasia share many common symptoms and causal factors, the progression and onset period greatly differ. The symptoms of Friedreich's ataxia often appear at a much later stage of childhood (15-20 years of age). Spinocerebellar ataxia: Also known as spinocerebellar atrophy or spinocerebellar degeneration, it is an ataxia that often doesn't show signs until adulthood and its symptoms usually appear between 25 to 80 years of age. Currently, based on their unique genetic abnormalities and pattern of inheritance (passing from parents to children), spinocerebellar ataxias are classified into 36 different types (SCA1 to SCA36). Further, based on their distant characteristics and association with defective genes, the 36 different types of SCA are grouped into the following subtypes: Subtypes of spinocerebellar ataxia Autosomal dominant cerebellar ataxia type I - includes syndromes like SCA1-SCA4, SCA8, SCA10, SCA12-SCA23, SCA25, SCA27, and SCA28. Autosomal dominant cerebellar ataxia type II - includes SCA7. Autosomal dominant cerebellar ataxia type III - includes SCA5, SCA6, SCA11, SCA26, SCA29, SCA30, and SCA 31. Idiopathic late-onset cerebellar ataxia (ILOCA): Also known as Multiple System Atrophy (MSA), it refers to conditions for which the exact cause and medical reason for why a person develops ataxia are not clearly understood and identified. Early Symptoms To Watch Out for Spinocerebellar Ataxia Spinocerebellar Ataxia Symptoms depend largely on the severity and stage of the condition. Some general symptoms of ataxia include Poor/decreasing control in mind-muscle coordination Slurred speech Difficulties in performing day-to-day fine motor activities Headache Dizziness Visual impairment Involuntary rapid eye movements Unsteady/difficulties in walking movements Loss of sensation Possible Causes Behind Spinocerebellar Ataxia Spinocerebellar ataxia happens largely due to defect or damage to the parts of the brain (cerebellum) that controls muscle coordination. Further, defects or mutations in genes responsible for the proper functioning of the nerve cells in the brain can also trigger and lead to spinocerebellar ataxia. Based on the nature of the causing factors, researchers and doctors have categorized ataxia into three major groups, namely, Acquired causes: This includes external factors that cause ataxia. Some potential external causes include Head injury/ trauma Adverse side effects of certain medications Thyroid problems Exposure to infection Cancer or other underactive illness affecting brain function Hereditary causes: The hereditary causes include defects/mutations in genes that lead to abnormal functioning of nerve cells in the brain. Based on their mode of inheritance (passage from parents to children) and certain other characteristics, they are mainly classified into two types, namely Autosomal Recessive: The affected individual gets defective genes from both the parents (mother and father). Ataxia-telangiectasia and Friedreich's ataxia are generally inherited in this way. Autosomal Dominant: The presence of a single copy of a defective gene (either from the mother or father) is enough to cause an ataxia condition. Many cases of spinocerebellar ataxia are inherited in this way. Degenerative causes: Age-related wear and tear on muscles and spinal discs, presence of other underactive illnesses such as high blood pressure, and internal haemorrhage are included under degenerative causes. How Is Spinocerebellar Ataxia Diagnosed? The diagnosis of spinocerebellar ataxia primarily targets evaluating the regions of the affected/ damaged areas of the cerebellum and analysing the type of genetic defect present in the individuals. Some standard diagnostic techniques used include Magnetic Resonance Imaging (MRI) scan of the brain Non-Invasive Computed Tomography (CT) scan to analyse the shrinkage in the brain Blood test to determine other associated illnesses Modern genetic testing to confirm hereditary related gene defects The DNA tests for Spinocerebellar Ataxia [SCA tests] further help to diagnose and classify the ataxia into various types. What Can Put You At a Risk Of Developing Spinocerebellar Ataxia? Many health conditions and factors can put you at risk of developing ataxia. Some possible risk factors of ataxia include Exposure to viral infections such as coxsackie virus, chicken pox, Epstein-Barr, coronavirus, or Human immune virus (HIV) Bacterial infections such as Lyme disease Vitamin and nutrient deficiencies Alcoholism Overuse of certain medications Presence of underlying blood clots, haemorrhage, or obstruction of a blood vessel What Are The Treatment Options Available For Spinocerebellar Ataxia? Currently, there is no effective or specific treatment for spinocerebellar ataxia (SCA). However, in general, the treatment for SCA usually focuses on alleviating the signs and progression of the condition. Some common treatments and strategies for managing the signs of SCA involve Physiotherapy to improve balance and mobility Speech therapy to improve slurred speech and swallowing problems Medical devices to assist in performing fine motor skills and self-care activities Medication to relieve symptoms such as tremors, depression, stiffness, muscle pain and sleep disorders What Is The Prognosis And Outlook For Individuals With Spinocerebellar Ataxia? Medical advancements and modern technologies are not adequately successful in preventing spinocerebellar ataxia. However, accurate diagnostic technologies are available to identify the defective genes in SCA patients. The next step is to correct those defects using gene therapy technology. The use of gene therapy technology is still in its infancy. However, the future holds promising results and outlook in treating SCA. CTA: Getting a Spinocerebellar Ataxia Type 12 (SCA12) DNA test along with other screening methods can increase the odds of early detection. The early detection of spinocerebellar ataxia may lead to better management, positive outcomes and longer survival in the patient.
Knowing Multiple Sclerosis Risk Factors and Life Expectancy: The Disease Prognosis, Diagnosis, and Treatment
A chronic illness affecting the Central Nervous System, Multiple Sclerosis impacts the entire human body. The symptoms of this disease are thus not located to any specific organ or region but instead can be observed throughout the body. Although not a fatal disease, Multiple Sclerosis impacts several organs and interrupts with their everyday functions. Multiple scar tissue, or lesions, formation in the spinal cord, brain and optic nerves’ nerve fibre is the most observable and detectable symptom of this auto-immune disease whose primary cause is not known. It is thus even more crucial to keep disease diagnosis scheduled and get assessed regarding what is Multiple Sclerosis life expectancy based on disease advancement. The illness is progressive and leads to body degeneration over the years. Women are at 3x the risk of developing Multiple Sclerosis. However, a cure for this degenerative disease has not been found yet. The treatments and medications thus mostly address the affected organs, tissues, or functions only and not the cause of such degeneration. Here’re some essential details related to the Multiple Sclerosis life expectancy, risk factors, and diagnosis that can be useful to know for a proactive approach to its prevention, identification, and impacts. Causes and Risk Factors of Multiple Sclerosis Although the primary cause for Multiple Sclerosis development is not known, several factors have been identified that influence or aggravate this medical condition. The Central Nervous System impacts the body’s movements, cognition, speech, sensation and memory functions which can be impacted. The control is achieved via electrical signals exchanged between the brain and the millions of nerve cells in the body. The nerve fibre of these cells is protected by a myelin sheath layer. This sheath also regulates brainwave signalling to the targeted cells, tissues, or organs. However, if a person is affected by Multiple Sclerosis, their faulty auto-immune response ends up attacking and damaging the protective myelin sheath. Such an error in the body’s immune system compromises the sheath’s ability to communicate the signals. Such damage can result in symptoms causing debilitation of body functions such as fatigue, slurred speech, weakness in the muscles, and coordination issues like trouble walking. As the impact of Multiple Sclerosis varies from one physiology to another, the severity levels and progression rate of this disease are different too. The risk factors that may aggravate the chances of the human body developing this disease can be primarily identified under four categories, which include - The body’s immunity: The immune system’s faulty targeting of the body’s foreign objects results in a damaged myelin sheath of the nerve fibre. The illness is thus also identified under the auto-immune diseases’ category. Genetic build: Multiple Sclerosis also has a genetic root. Individuals who have a close relative, like a sibling or a parent, affected by the disease carry a higher chance of developing it than others. However, it is not yet identified as a hereditary disease. Possible infections: Certain types of viral infections are known to aggravate the risk of developing Multiple Sclerosis. Some of them include EPV, VZV, HHV-6, CMV, and HERVs. Environment of the person: Some environmental aspects are also identified to aggravate the risk of developing the illness, such as air pollution, low or limited exposure to Vitamin D, and exposure to certain organic solvents. Some other risk factors that contribute to Multiple Sclerosis development or aggravation include obesity during childhood or adulthood, smoking habits, age, and gender. A study of the disease pattern has also suggested the contribution of geographic gradient towards the disease’s development. People living farther from the equator are more likely to carry the risk of developing it. Multiple Sclerosis Disease Prognosis and Diagnosis As several symptoms of Multiple Sclerosis mimic those of other medical conditions, diagnosis of the disease becomes tough. However, there are some patterns and types that can confirm the disease’s presence in one’s body after due examination and conducting necessary tests. Also, it takes multiple tests to identify its presence. It may not be possible to determine if you have been affected by Multiple Sclerosis or not after only one attack of the possible symptoms. Some common ways to diagnose Multiple Sclerosis are listed below. An examination of neurological factors for possible symptoms such as issues with the vision, movement of the eyes, limb strength, reflexes, and coordination can suggest nerve fibre damage, leading to the symptoms. Detailed imaging of the Central Nervous System via an MRI scan can also help identify the disease’s prevalence. The various types of evoked potential tests can track the different eye movements that the body undertakes as a reflex to various light waves, creating a brainwave map for possible neural disconnect or abrasion. CSF through lumbar puncture to detect intrathecal production of IgG antibody and oligoclonal band can help diagnose the disease. Several blood tests are also conducted to rule out any other possible causes of the observed symptoms. Anti myelin associated glycoprotein (MAG antibody) can be detected in serum of patients suffering from MS. The disease’s prognosis should be done through an identification of various symptoms as per the type of Multiple Sclerosis that the individual suffers. Some factors that can help determine the type of this illness include - Symptom severity Rate of disease progression The time gap between two consecutive flares Type of treatment administered Possible comorbidities The Multiple Sclerosis Types Identifiable Based on This Prognosis are Characterised as - Primary Progressive Multiple Sclerosis Secondary Progressive Multiple Sclerosis Relapsing Remitting Multiple Sclerosis Clinically Isolated Syndrome Treatment of the disease can follow various available paths as well, such as through medications for relapse management, DMTs or Disease Modifying Therapies, mental health counselling, and physical rehabilitation. The advanced Multiple Sclerosis life expectancy can, however, be a matter of concern needing critical medical care. Life Expectancy Among The Affected Patients Even though Multiple Sclerosis is not a life-threatening, fatal disease, its progression can impact the overall Multiple Sclerosis life expectancy of the patient. While many patients affected by the disease carry a life expectancy that is close to normal. However, due to a widely varying disease progression scale, the predictability for an acute impact on Multiple Sclerosis life expectancy is low. Research suggests that two-thirds of the affected individuals carry up to two decades of their lives without any artificial support such as a wheelchair. In another study, the Multiple Sclerosis life expectancy has been found to reduce by up to 7.5 years in patients suffering from Multiple Sclerosis. If you suffer from any of the possible symptoms indicating the disease’s onset, it is best to consult a specialist medical professional for disease diagnosis. Since isolated cases of symptoms’ appearance may not reveal the integral nerve fibre damage, one must be vigilant and cautious regarding repeating symptom appearances and undergo suitable treatment for the elimination, or at least reduction, of its prognosed symptoms. Metropolis Healthcare Ltd. offers both CSF and serum based tests for diagnosis of the disease. Make sure to consult with your doctor before going for these tests.
Coping With a Lung Cancer Diagnosis
The doctor will screen you for lung cancer if a tumour-like growth in the lungs is detected during a CT (Computed Tomography) scan. Lung cancer has general symptoms too, including: Persistent cough Blood in the sputum Loss of appetite Weight loss for no reason Feeling tired Breathlessness Pain in chest But remember, not every type of lung cancer has symptoms. A positive cancer diagnosis can occur without symptoms too. Cancer is the most dreaded of all ailments. Diagnosis of lung cancer can come with social stigma too since many might wonder if you have a smoking habit which caused the cancer. Around 95% of lung cancer patients have a history of smoking. This article has a list of tips that can help you cope with the emotional and physical toll your lung cancer diagnosis can bring. What is ALK-Positive Lung Cancer? ALK is an abbreviation for anaplastic lymphoma kinase. ALK is the gene in the body present from the embryo stage. The gene helps develop our nervous system and the gastrointestinal tract (gut). ALK-positive lung cancer occurs when there is a mutation or a misarrangement in the ALK gene. When such mutation occurs in the DNA of lung cells, they grow abnormally and multiply into more abnormal cells, causing cancer. These cancer cells start in the lungs and may eventually spread to other parts of the body, making it stage 4 (secondary or metastatic) cancer. The ALK-positive lung cancer forms around 4% of non-small cell lung cancers (NSCLC). There is a higher percentage of ALK-positive lung cancer in patients younger than 55 years of age and never smoked. Confirmation of ALK-Positive Lung Cancer When our individual DNA is being ‘written’ and if there is a typing error, the cells receiving the wrong information will grow into a tumour. Diagnostic tests such as biomarker testing look for such typing mistakes. The ALK-positive lung cancer can be further confirmed by testing your blood samples and a tumour biopsy. The procedures involve: Microscopic analysis of chromosomes Immunohistochemistry analysis of proteins Next generation sequencing Liquid biopsy The treatment is decided once the type and stage of cancer are determined. Remember that ALK-positive lung cancer stage 4 has the most treatment implications. How to Cope with Emotions After a Lung Cancer Diagnosis? A doctor saying “you have lung cancer” can be the most feared sentence you have ever heard. Your immediate reaction might be to go numb, hopeless, irritated, angry, or confused as to why it has happened to you, especially when you can’t think of anything you have done personally to get harmed in such a way. It is okay to experience these emotions. As you come to terms with the diagnosis, it is okay to experience extreme emotions at such a time. While trying to find treatment centres and experts who can help with medical options, here are a few supportive tips for coping with the lung cancer diagnosis. Try to put aside anything that is not urgent or important. Use this time to focus only on getting through this challenging time emotionally. Accept that you are not alone. Many people will help you in this journey. Seek help. Find out who can exactly help you and how. Keep away the guilt. Lung cancer patients often think that they made this happen. They start connecting some bad habits, especially smoking with cancer. However, if you let the guilt in, your tensions and worry will only increase. Share your version with your family and friends. They will provide practical and emotional support. Be kind to yourself. You may feel low and tired at times, but be extra kind and prepare yourself for the treatment. It is natural to worry about the treatment. Write down your fears about the treatment and its side effects, and discuss them with your doctor. When you are ready, start gathering facts from an expert oncologist about the type of cancer you have and the possible treatment options. Think ahead and organise your life, so you are set to receive the treatment. At home and office, take help to manage your routine. Be with those who lift your spirit. A cancer diagnosis can change your relationships. Those who love you are equally affected by your diagnosis. Hence sharing this news with them may be the hardest for you. Remember to stay close to every person you love. If they are present during the treatment and caring process, make sure they take enough breaks for themselves. Talk to your healthcare team and know more about your treatment and what to expect from it. This will help you plan while keeping the treatment goals in mind. Manage your symptoms with the help of a nurse or a professional caregiver. Breathlessness and pain can be expected if you have lung cancer. Get into a healthy lifestyle with home-cooked food. Make sure that you eat nutritious meals at regular intervals. Move around as much as you can afford to do, as it will keep your energy levels up. Avoid strenuous exercises. If you are bedridden or bound to a wheelchair, discuss with your doctor or physiotherapist about exercises. Learn yoga and meditation to help you relax and sleep peacefully. Getting enough sleep daily will keep your emotional energy up. Join a support group to share your emotional problems with people who have gone through similar experiences. Whether the diagnosis is made during the early stages of ALK-positive lung cancer or near stage 4, and whatever may be the life expectancy, remember you are not alone. Take necessary support, plan your days and face the treatment head-on so that you can beat cancer. Your doctor may advise you to go for ALK for NSCLC (IHC reflex to FISH) Test. IHC is immunohistochemistry, and FISH is Fluorescence in situ hybridisation. This test is approved for the diagnosis of lung cancer and to check the status of ALK. In the case of an ALK-positive test, your doctor may start targeted therapy such as the treatment of tyrosine kinase inhibitors. Book your test now and get accurate and quick results at Metropolis.
Thrombophilia in Pregnancy: Screening, Prevention and Treatment
Thrombophilia means the blood can easily form clots, sometimes even without injury. This is also called hypercoagulability. It occurs when the body generates too much clotting protein. Normally, when you get a cut or are injured, blood clots stop the bleeding. Later, your body breaks down the clot when it’s not needed anymore. When you have thrombophilia, the body makes too many blood clots or the old clots remain intact. In thrombophilia, the oxygen supply is cut off to other tissues and organs. It may result in a blood clot in the leg, deep vein thrombosis (DVT) or a pulmonary embolism. It can also result in a heart attack or a stroke. Usually, the condition is asymptomatic unless a clot appears. Hence, estimating how many people are affected by thrombophilia is difficult. Thrombophilia can be inherited, or it can develop later in life. Types of Thrombophilia 1. Inherited types Factor V Leiden is a common genetic form that primarily affects Europeans. Genetic types of thrombophilia may have a high risk of multiple miscarriages. Even with this mutation, most women have normal pregnancies. Other inherited types include: Hereditary antithrombin deficiency Protein C deficiency Congenital dysfibrinogenemia Protein S deficiency 2. Acquired types Antiphospholipid syndrome is one of the most common acquired types of thrombophilia. About 70 per cent of those affected are women. The antiphospholipid syndrome may increase the risk of pregnancy-related complications such as: Preeclampsia Miscarriage Stillbirth Fewer birth weights Other causes of acquired thrombophilia include: Prolonged bed rest Cancer Acquired dysfibrinogenemia Traumatic injury Symptoms of Thrombophilia Thrombophilia doesn’t have any symptoms unless you have a blood clot. However, some symptoms include: Chest pain Lightheadedness Shortness of breath Pain in the upper back Fainting Dizziness Dry cough Coughing blood Recurrent miscarriage What are the Risk Factors of Thrombophilia? The following reasons can make you more susceptible to thrombophilia. Being overweight Pregnancy Smoking Cancer Diabetes Heart problems Prolonged bed rest Taking birth control pills Taking hormonal replacement therapy Old age Who Should be Tested for Thrombophilia? You should get tested for thrombophilia if: A family history of blood clots Blood clots in unusual areas You went through multiple miscarriages Thrombophilia in Pregnancy More than half of thromboembolic events during pregnancy are due to thrombophilia. Recurrent loss of pregnancy or miscarriage occurs in about 5% of women of reproductive age. Hereditary and acquired thrombophilia are responsible for approximately 50 per cent of thrombotic events diagnosed during pregnancy and postnatal. Some inherited thrombophilia factors like factor V Leiden mutation, protein C deficiency, protein S deficiency, antithrombin deficiency and prothrombin G20210A mutation may cause thromboembolism in pregnant women. Inherited thrombophilia is associated with fetal growth restriction. Studies have linked thrombophilia to adverse pregnancy outcomes, resulting in preeclampsia and intrauterine growth restriction. It is assumed there might be a maternal predisposition to clotting that leads to thrombosis in the placental vasculature, eventually restricting the supply of oxygen and exchange of nutrients, resulting in fetal growth restriction. The following symptoms can accompany thrombophilia: Repeated miscarriages Preeclampsia Intrauterine growth restriction Neonatal fulminant purple HELLP syndrome Stillbirth Diagnosis for Thrombophilia Your doctor will recommend blood tests to measure the levels of clotting proteins. They will look for gene abnormalities and certain antibodies. These tests can decide your treatment choices, such as the type of blood thinner you should take and the duration. A screening test can help determine how often you will get blood clots and investigate whether any of your family members have thrombophilia. To understand the importance of test results, it is advisable to get help from a genetic counsellor. What are Thrombophilia Profile Tests? Thrombophilia does not cause visible symptoms. Even if you have the condition, you will not know unless you have a blood clot. Screening for thrombophilia includes: Antithrombin Lupus anticoagulant Prothrombin gene mutation Protein C Protein S anti-cardiolipin antibodies Factor V Leiden anti-β-2-glycoprotein-1 antibodies Icd 10 Thrombophilia Test is performed to check early haemorrhage before completion of 20 weeks of gestation. How is Thrombophilia Treated? You may not need treatment for thrombophilia until you get a blood clot or have a high risk of getting one. You may need a blood thinner as a long-term treatment if you have the antiphospholipid antibody syndrome form of thrombophilia. Women should avoid birth control pills that have oestrogen in them. Instead, you are advised to take progestin-only pills. Your doctor may advise avoiding hormone replacement therapy for menopause symptoms. Medications may include anticoagulants, such as warfarin or heparin, that will show the effect in a few days. For immediate blood clot treatment, you can use heparin for a better outcome. It is a fast-acting drug that can be used with warfarin. How to Prevent Blood Clots due to Thrombophilia? You can prevent unwanted blood clots by changing your lifestyle. You can: Maintain a healthy weight Stop smoking Exercise regularly Avoid sedentary lifestyle Get treatment for conditions that can cause thrombophilia Take your blood thinner medication regularly. Watch for any signs of bleeding, such as nosebleeds and blood in urine and stool. If you have any concerns with your blood thinner, immediately inform your doctor. People with cancer should always be aware of the latest screenings and medications because they have a high risk of developing blood clots. Closing thoughts Inherited thrombophilia cannot be prevented, whereas acquired thrombophilia can be prevented but not completely. To decrease your chances of developing blood clots, look for the warning signs and get treated immediately. Some people have thrombophilia but never develop blood clots, nor do they need treatment. But in some cases, doctors may recommend the long-term use of blood thinners and regular blood tests. Thrombophilia can be managed successfully by regular follow-ups with your doctor.
Myositis Symptoms and Possible Treatment
The meaning of myositis is very simple. It is a group of conditions causing muscle weakness, pain and inflammation resulting in complete damage. The problem starts gradually but can take varied forms. An unusual rash is sometimes the first sign of this condition. Sometimes the patients might start falling or tripping more frequently. Most often, patients misunderstand myositis symptoms as signs of old age. But the condition is real and can even become serious if not taken care of. Significant causes of myositis include injury, infection, drug side effects and autoimmune conditions. The treatment for this problem can differ as per the cause. Myositis Types and Symptoms The different types of myositis include: Dermatomyositis - affects women and children. Is associated with rash. Polymyositis - affects women between 30-60 years most often Necrotizing Myopathy Juvenile Myositis Sporadic Inclusion Body Myositis (IBM) You need to know more about Sporadic inclusion body myositis symptoms as it is the most common form of myositis found in people above 50 years of age. Major symptoms of this myositis type include difficulty climbing stairs or walking. Muscle weakness and inflammation are the main symptoms of myositis. This weakness might be noticeable, or you can find it only through testing. Myalgias or muscle pain may or may not be present. Polymyositis, Dermatomyositis and other inflammatory myositis cases cause muscle weakness that worsens over time. Muscle weakness generally affects large muscle groups, such as the shoulders, neck, back and hips, and muscles on both sides are usually affected. Muscle weakness from the condition can result in falls and also make it difficult for patients to get up after a fall or from a chair. Other common signs of this inflammatory condition are: Fatigue Rash- In dermatomyositis Difficulty swallowing - In IBM Thickening of the skin on the hands Pain in the muscles People with myositis caused due to a virus can have the signs of viral infection, like fever, runny nose, sore throat, diarrhoea, nausea and cough. However, the signs of viral infection usually go away days or even weeks before the signs of myositis begin. Myositis Diagnosis Metropolis Healthcare Ltd. offers an immunoblot test to diagnose the disease. Test can detect up to sixteen different types of auto-antibodies which are responsible for the condition. Myositis Treatment Myositis treatment can vary as per the cause of the condition. However, basic treatment for the condition includes a blend of exercise and drugs. Medicines are the first choice of treatment for the condition. High doses of steroids are administered in the beginning. These are given as injections or tablets, and they help reduce inflammation and muscle pain along with the feeling of being unwell. Inflammatory conditions that cause myositis are treated with medicines that suppress the immune system. These include: Azathioprine (Imuran) Prednisone Methotrexate There is no specific treatment for myositis caused due to a viral infection. Myositis caused by the drug is treated by stopping the drug. People suffering from myositis due to statin drugs usually experience muscle inflammation that subsides within just a few weeks of stopping the drugs. Most of the time, the medicines mentioned above are unable to treat myositis leading to more severe cases that require the use of treatments like: Immunoglobulins Immunoglobulins are the antibodies collected through blood donations offered by healthy individuals. These can stop the immune system from attacking the tissues in the body. It is offered in a hospital setting and is even repeated if required. It is likely for people to feel a bit unwell during this treatment. Biological therapy Biological therapies treat myositis by blocking targets in the immune system that lead to inflammation. Even the most serious cases of myositis respond to proper treatment, but patients might require life-long medicines to keep the condition in control. Physiotherapy and Exercise Rest is vital in an active myositis condition. And once the problem is better, exercise regularly to improve its symptoms and overall health. Essentially, aerobic exercises that make the heart beat faster and a person breathe more heavily are crucial to improve stamina and regain muscle strength. Do such exercises under a physiotherapist’s supervision who usually offers tailored programs as per the requirement of the patients. Myositis patients should never engage in strenuous exercises. Kids with juvenile Dermatomyositis require more energetic physiotherapy to permanently avoid bending knees and other joints. Myositis patients can recover properly, but not all can recover completely. Even for the ones who make a good recovery, it might take several months to see the improvement. That’s because treatment takes time to help the body repair the damaged muscles, resulting in tiredness among the patients. The key is to continue exercises, which can help with different aspects of myositis. The Bottom Line So, this is all about myositis symptoms and treatment. Myositis can present itself in varied forms affecting the entire body, not just the muscles. Therefore, proper diagnosis of the condition is not always possible. However, an all-inclusive assessment can make it easier to treat myositis patients. Besides that, you can opt for Myositis Profile-IgG Test to detect the particular autoantibodies for identifying myositis.
What You Should Know About Multiple Sclerosis (MS) Symptoms In Female
The last thing anyone should worry about is a potential MS diagnosis. However, it's essential to be aware of the symptoms to get help and determine if you should see a doctor. In this post, we'll outline the most common multiple sclerosis symptoms in females and provide tips on how to deal with them. Whether you're a woman recently diagnosed with MS or just concerned about your health, read on for everything you need to know. What is Multiple Sclerosis? Multiple sclerosis is a disease that affects the nervous system. It's caused by damage to the myelin sheath – a protective membrane covering nerve cells in the brain and spinal cord. This leads to lost function or paralysis in one or more limbs, difficulty with movement, vision problems, and hearing problems. Multiple sclerosis symptoms in females can often be mistaken for other conditions, such as anxiety or depression. Please seek out professional help if you believe you might be experiencing these MS-related symptoms: 1. Fatigue and/or weakness: This is perhaps the most common symptom of MS and can make everyday tasks quite difficult. If your energy levels are consistently low, it may be a sign that you need to see a doctor for an evaluation. 2. Sensitivity to light and sound: Many people with MS experience significant problems with their vision (including seeing patterns not apparent to others) and hearing. If you're constantly feeling winded or struggling to hear, it may signal that you need help. 3. Trouble with balance and coordination: MS can cause problems with balance and coordination, making everyday tasks difficult – such as walking down the street or getting up from a seated position. 4. Difficulty speaking clearly: Many people with MS find it hard to speak clearly due to impaired speech production (known as dysarthria). 5. Difficulty swallowing: People with MS often experience difficulty swallowing, leading to poor nutrition and malnutrition. If you've noticed that your food doesn't seem to be staying down or you have trouble drinking liquids, it may be a sign that you need to see a doctor for an evaluation. 6. Joint pain: Joint pain is a common symptom of MS and can often be severe. If you're experiencing constant joint pain or if it becomes particularly bad during certain activities (like exercise), it may be time to see your doctor for an evaluation. 7. Memory problems: Many people with MS experience significant memory problems, making everyday tasks – such as shopping for groceries or getting on an aeroplane – quite tricky. If you find that your memories are slowly deteriorating or have trouble concentrating for longer durations of time, it might be time to seek professional help. Causes of Multiple Sclerosis MS is an umbrella term that refers to various diseases and conditions. While the precise cause of MS is unknown, researchers believe that environmental and genetic factors may cause it. Risk Factors of Multiple Sclerosis There are several risk factors associated with developing MS, including: 1. Being female 2. Having a family history of MS 3. Having had multiple sclerosis before 4. Exposure to certain viruses or toxins 5. Having an autoimmune disorder 6 . Inability to metabolise certain medications correctly 7. Early signs of ageing Diagnostic Criteria for MS The Diagnostic Criteria for MS, published in the Annals of Neurology, are a set of criteria that doctors use to diagnose MS. The following three symptoms must be present together for at least six months and must cause significant disability: 1. Intense or painful neurological symptoms (at least 2 out of 7) 2. Eye problems, such as blurred vision or blindness 3. Swollen lymph nodes Prevention of Multiple Sclerosis There is no known cure for MS, but many ways exist to prevent its onset and progression. Some essential lifestyle habits that you can adopt are: 1. Keeping your body healthy by eating a balanced and healthy diet 2. Getting regular physical activity 3. Avoid excessive alcohol and tobacco use Treatment of Multiple Sclerosis There is currently no cure for MS, but various treatments may help improve the quality of life. Some common treatments include: 1. A combination of physical and cognitive therapy to help restore mobility and function 2. Medications to relieve symptoms such as pain, inflammation, and bladder control problems 3. Surgery to remove scar tissue or tumours from the brain or spinal cord 4. Intravenous immunoglobulin (IVIg) to help reduce the severity and frequency of attacks 5. Social support programs help people with MS connect with other community members. Complications MS can lead to a wide variety of complications, some of which are very serious. Some common complications include: 1. Cognitive decline and memory loss 2. Depression or anxiety 3. Difficulty walking or using stairs 4. Cranial nerve damage, such as problems with vision or hearing 5. Severe bladder and bowel problems 6. MS- Related paralysis Conclusion Even though women with MS often show no symptoms, they can still be at risk of developing the disease. This is why awareness and early detection ensure a healthy future. While no two people experience MS in the same way, know your body – talk to your healthcare providers about any concerns you may have about managing your symptoms or treating your disease. A Multiple Sclerosis Profile and a Maxi Test are the most effective diagnostic tools for the condition. Knowing what to look for and taking action at the right time can get you one step closer to living a healthier life!
Acute Leukaemia Lymphoma - Symptoms, Causes, Risk Factors and Treatment
Acute leukaemia is a severe disease that progresses rapidly. Its symptoms show within weeks after the formation of leukaemia cells, because of which, patients need immediate treatment. Acute lymphocytic leukaemia is most common in children, but adults may also develop the condition. What is Acute Leukaemia Lymphoma? Acute leukaemia lymphoma is a cancer that affects bone marrow and blood. It starts in the immune system's infection-fighting lymphocytes. Due to this disease, lymphocytes develop uncontrollably. They spread to the spleen, brain, spinal cord, lymph nodes, liver, and testicles (in males). Acute Leukaemia Symptoms Acute leukaemia symptoms are as follows: Fever Infections that occur frequently Gum disease causing bleeding Aches and pain in the bones Severe nosebleeds Lymph node lumps in the neck, abdomen, armpits, or groin Shortness of breath Weakness, fatigue, or energy loss Acute Leukaemia Causes The DNA of a cell contains instructions that the cell must follow. It instructs the cell to grow and die at a certain rate. Acute lymphocytic leukaemia develops when a bone marrow cell's genetic material (DNA) develops mutations and drastically speeds up cell production. The bone marrow produces immature cells that grow into leukemic lymphoblast. These abnormal cells cannot function properly and affect healthy cells. When should you see a doctor? Symptoms of acute lymphocytic leukaemia are similar to those of flu. Flu symptoms, however, gradually go away. If your symptoms do not go away as soon as you expect them to, book an appointment with your doctor immediately. Risk factors of Acute Lymphocytic Leukaemia? Risk factors for acute lymphocytic leukaemia are as follows: 1. Radiation Exposure There is an increased risk of acute lymphocytic leukaemia among those exposed to high radiation levels, such as nuclear reactor accident survivors. 2. Genetic Disorders Acute lymphocytic leukaemia is more common in people with Down syndrome and other genetic disorders. 3. Previous Treatments for Cancer Children and adults who have undergone certain forms of chemotherapy and radiation treatment for other types of cancer are more prone to develop acute lymphocytic leukaemia. How do Doctors Diagnose Acute Leukaemia? 1. Bone Marrow Test In this test, doctors take a sample of bone marrow from the breastbone or hipbone using a needle. The lab runs tests to identify leukaemia cells. Based on their size, shape, and other genetic or molecular features, medical professionals in the lab categorise blood cells into different categories. Additionally, they analyse the cancer cells for changes and determine if the leukaemia cells came from B or T lymphocytes. Doctors then use this information to create a treatment plan. 2. Imaging Tests If cancer has progressed to the brain and spinal cord or other organs, imaging tests like X-ray, CT scan, or ultrasound scan can help in the diagnosis. 3. Spinal Fluid Test Doctors collect spinal fluid around the brain and spinal cord in a lumbar puncture test or spinal tap. They examine the sample to determine whether cancer cells have spread to the spinal fluid. 4. Blood Tests Blood tests may show excessive or insufficient white blood cells, insufficient red blood cells, insufficient platelets, or immature bone marrow blast cells. Doctors use this information to determine your prognosis and treatment options. What are the Treatment Options for Acute Leukaemia? The treatment options for acute leukaemia are described below. 1. Targeted Therapy Targeting certain proteins, genes, or other elements that support cancer growth is part of the relatively new approach known as targeted therapy. Blocking these factors may slow or stop the spread of cancer. Although the treatment should have fewer side effects than chemotherapy since it targets a particular disease, adverse effects may still develop. 2. Radiation Therapy Doctors use X-rays in radiation treatment to kill cancer cells. If the disease has spread to the nervous system, a doctor may recommend radiation therapy. 3. Bone Marrow Transplant Doctors perform a bone marrow transplant as consolidation therapy or to treat relapse. This procedure replaces the leukaemic bone marrow of a patient with healthy bone marrow from a healthy donor. Doctors use high doses of chemotherapy or radiation before a bone marrow transplant to kill any leukaemia-causing bone marrow. It is then necessary to replace the patient's bone marrow with the donor’s bone marrow. 4. Engineering Immune Cells to Fight Leukaemia Chimeric antigen receptor (CAR)-T cell therapy is a specialised therapy that includes extracting T cells from the immune system, modifying them to fight cancer, and then injecting them back into the body. CAR-T cell therapy may be an option for teenagers and children. Doctors may use it for consolidation therapy or treating relapse. 5. Chemotherapy Chemotherapy is a common therapy for children and adults. Doctors use chemotherapy for consolidation and maintenance. How can you Prevent Acute Lymphocytic Leukaemia? Avoid exposure to radiation, toxins like pesticides or benzene, and any form of smoking or tobacco use. Additional lifestyle changes, including maintaining an active lifestyle and consuming nutritious food, may help lower the chance of getting acute lymphocytic leukaemia. Conclusion Acute lymphocytic leukaemia is a kind of leukaemia that is a blood and bone marrow malignancy. Although it is a common type of cancer in children, its 5-year survival rate is close to 90%. Adults are more likely to experience the life-threatening effects of acute lymphocytic leukaemia than children. Radiation therapy, targeted therapy, and chemotherapy are available as treatment options. Patients of acute lymphocytic leukaemia need continuous follow-ups to ensure their condition is stable. It is recommended to get an acute leukaemia panel and bone marrow test to detect acute leukaemia early.



1707898101.webp)